About 13th Annual EU Medical Device Regulatory Affairs Conference 2022
Optimize your regulatory oversight with perspectives from notified bodies and industry peers. Speakers exchange practical methods to strengthen regulatory strategy, best practices for communicating with notified bodies and lessons learned in receiving approval with limited guidance. Improve patient outcomes and internal operations by gaining methods to achieve sufficient data for successful product submissions.
Foster a sense of collaboration and safely connect with peers and expert presenters at this intimately-sized program. Customize your experience and address your unique concerns during dynamic Q&A after each session. Our commitment to industry perspectives keeps the program focused on what matters and dynamic session types allow you to stay engaged.
When and Where
Event date
Oct 19 - 20, 2022
Event location
Belgium
Event type
Event fees
Organized by
-
Reviews
Reviews Awaited
Please Note: Reviews are typically collected post conclusion of the event to allow for a well-informed opinion. Please check again after some time.



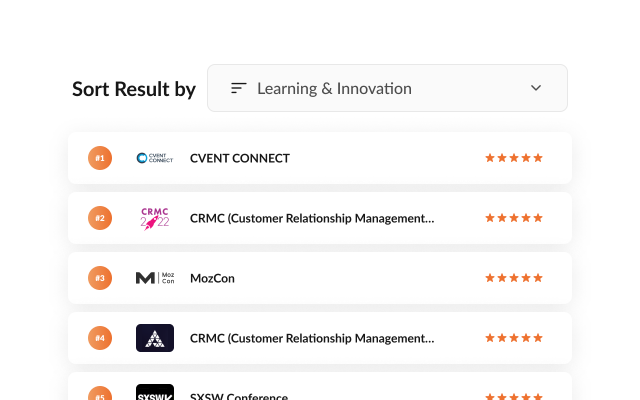
Choose your Goal
Select the priority e.g Networking or Learning that closely matches the objectives you would like to meet by attending an event.
Activate Reminder
Sign in or create your free account and click the bell icon to be notified of an upcoming edition for this event.
Receive Alert & Updates
Get timely notifications from Eventible before the start of the next edition. Never miss a must-attend event again!
Explore Upcoming Events
“No events listed. Please check back later!”
Explore Top Rated Events