About 3rd Annual Extractables & Leachables Summit 2023
When and Where
Event date
Mar 09 - 10, 2023
Organized by
Uventia Global s.r.o.
Event type
Event fees
Event location
Czech Republic
Event Snapshot
Audio Reviews (1)
Reviews
Greg Erexsononline | speaker
CEO & President, Greg Erexson Toxicology Consulting, LLC
Review Source
Review Source
Overall Online Experience
Eventible is a neutral platform, unaffiliated with organizers or partners. Reviews reflect attendees' personal opinions.
15th Mar '23
15th Mar '23
Andrew Feildenonline | speaker
Director Analytical Science, Bicycle Therapeutics
Review Source
Review Source
Overall Online Experience
Eventible is a neutral platform, unaffiliated with organizers or partners. Reviews reflect attendees' personal opinions.
16th Mar '23
16th Mar '23
Anurag Bhattacharjeeonline | attendee
Masters Student, OVGU Magdeburg
Review Source
Review Source
Overall Online Experience
0.000.00
Eventible is a neutral platform, unaffiliated with organizers or partners. Reviews reflect attendees' personal opinions.
15th Mar '23
15th Mar '23



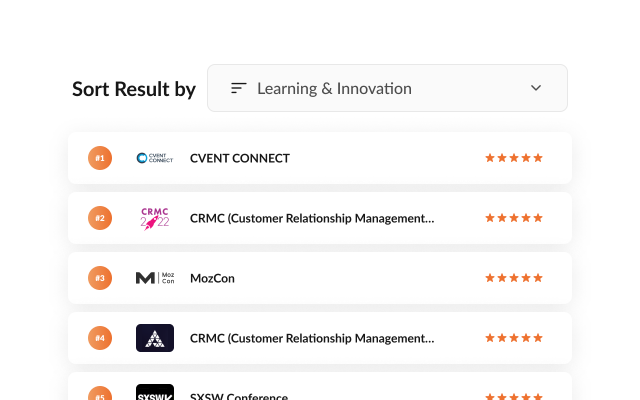
Choose your Goal
Select the priority e.g Networking or Learning that closely matches the objectives you would like to meet by attending an event.
Activate Reminder
Sign in or create your free account and click the bell icon to be notified of an upcoming edition for this event.
Receive Alert & Updates
Get timely notifications from Eventible before the start of the next edition. Never miss a must-attend event again!
Explore Upcoming Events
“No events listed. Please check back later!”
Explore Top Rated Events